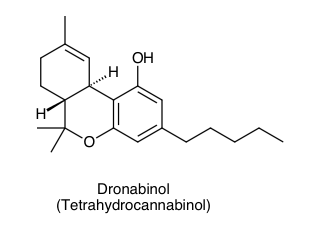
Thin-layer chromatography (TLC) of a sample of hashish (cannabis resin,... | Download Scientific Diagram

Study: Frequent Use of the Cannabis-Based Prescription Drug Associated with Opioid-Sparing Effects - NORML

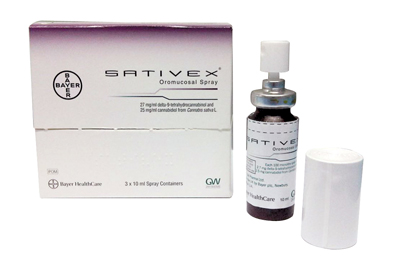
Canna Product 420 - canna_product_420 What is Sativex? Sativex® is a cannabis-based product classified as a Schedule 2, Part 1 (Class B1) controlled drug product under the Misuse of Drugs Act 1975.

Delta-9-Tetrahydrocannabinol/Cannabidiol (Sativex®): A Review of Its Use in Patients with Moderate to Severe Spasticity Due to Multiple Sclerosis | springermedizin.de
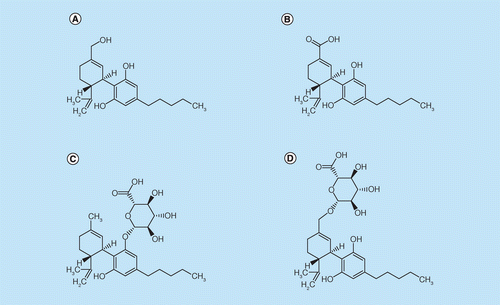
Techniques and technologies for the bioanalysis of Sativex®, metabolites and related compounds | Bioanalysis

Nabiximols (THC/CBD Oromucosal Spray, Sativex®) in Clinical Practice - Results of a Multicenter, Non-Interventional Study (MOVE 2) in Patients with Multiple Sclerosis Spasticity | Semantic Scholar