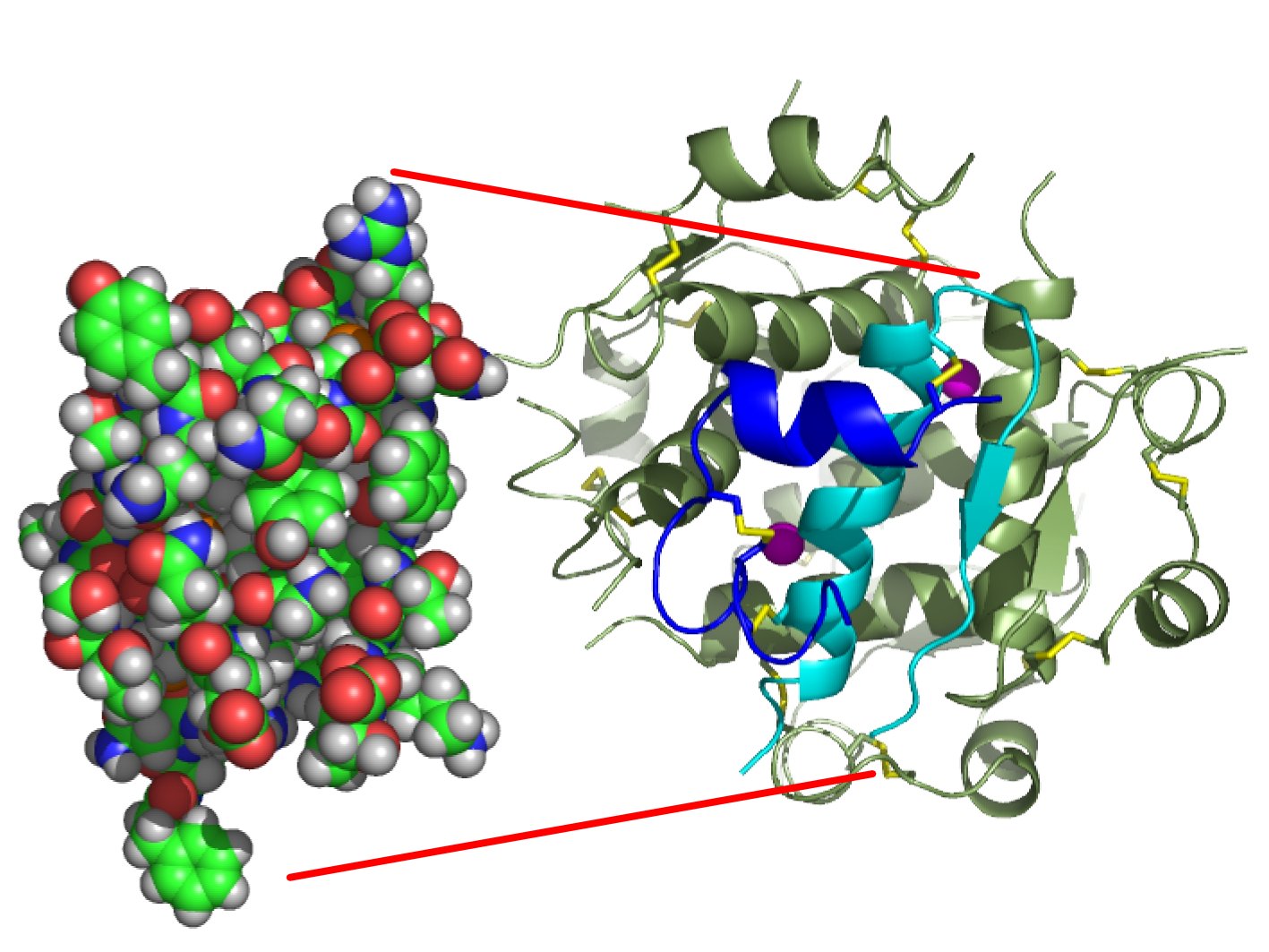
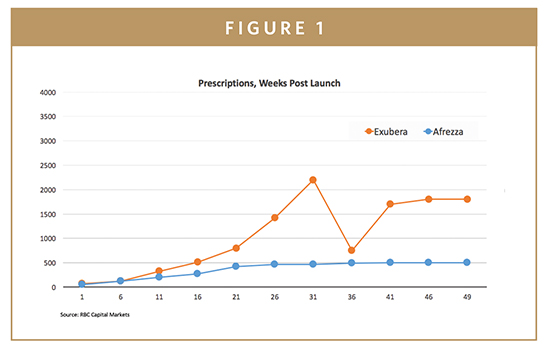
Future of Afrezza Inhaled Insulin: Interview with Michael Castagna, CCO of MannKind Corp. | Medgadget

Efficacy, safety, and patient acceptability of Technosphere inhaled insulin for people with diabetes: a systematic review and meta-analysis - The Lancet Diabetes & Endocrinology
Sanofi and MannKind announce global licensing agreement for Afrezza® ( insulin human) rapid-acting inhaled insulin - Drug Target Review

NRx Pharmaceuticals Partners with MannKind Corporation to Develop ZYESAMITM (aviptadil) Inhaler for Respiratory Conditions – Delco Times
![Diabetes without fear - Diabetes sem medo - Afrezza (human insulin [recombinant DNA origin]; (MannKind Corporation, Valencia, California, USA) is a drug-device combination consisting of pre-metered, single-use cartridges of TI inhalation powder Diabetes without fear - Diabetes sem medo - Afrezza (human insulin [recombinant DNA origin]; (MannKind Corporation, Valencia, California, USA) is a drug-device combination consisting of pre-metered, single-use cartridges of TI inhalation powder](https://lookaside.fbsbx.com/lookaside/crawler/media/?media_id=1395445637138116)