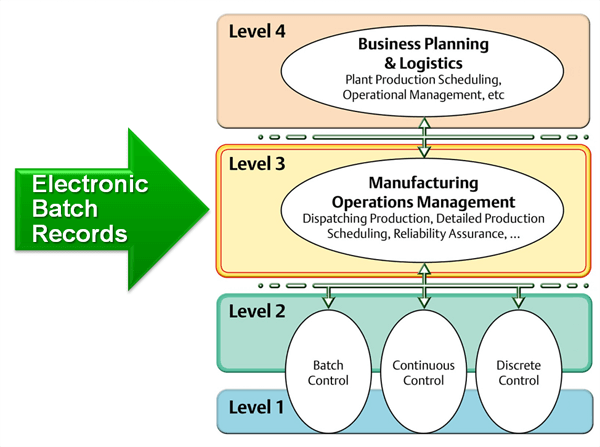
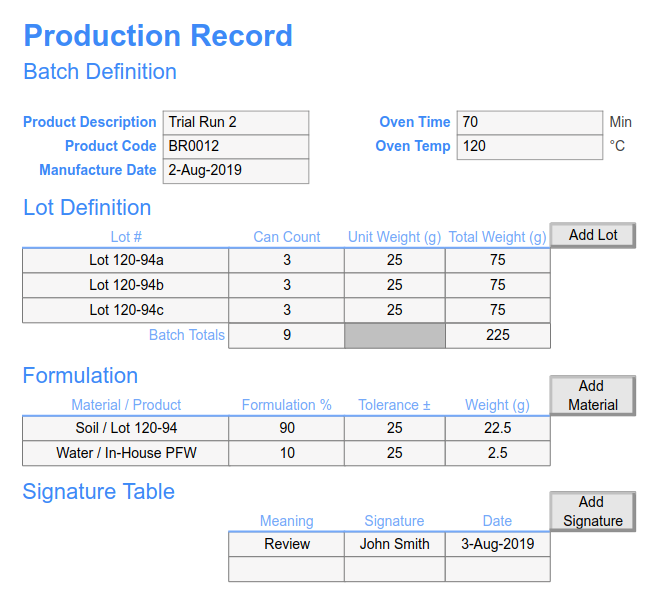
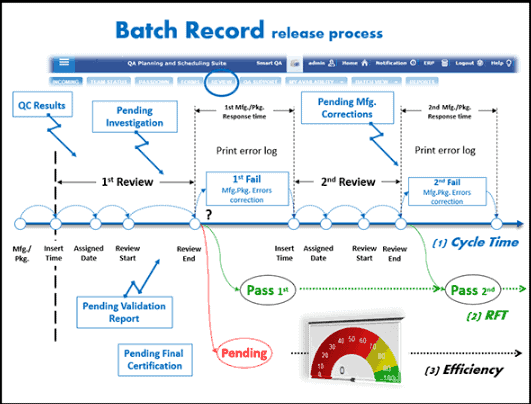
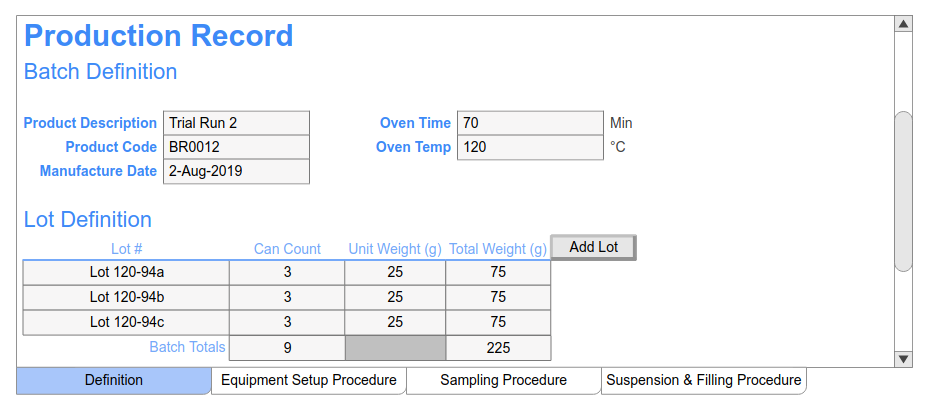
Batch Record MBR-007 Supersedes: None Version: 1.0 Title: CipA1 – Ciprofloxacin Hydrochloride tablets 200mg (F-7007) Effectiv

ASEAN-TMHS-GMP-Training-Chapter-5-Annex-5-Sample-Batch-Manufacturing-Record.doc - COMPANY NAME Department : Production BATCH MANUFACTURING RECORD Title | Course Hero
Corso di Laurea Magistrale in Chimica e Tecnologia Farmaceutiche – E25 Fabbricazione Industriale dei Medicinali – 4 CFU

Batch Record MBR-046 Supersedes: New Version: 1.0 Title: Fexo A – Fexofenadine HCl tablets 60mg (F-7046) Effective: Page 1 of




-en.jpg?Status=Master&sfvrsn=ce9a7fcc_0)