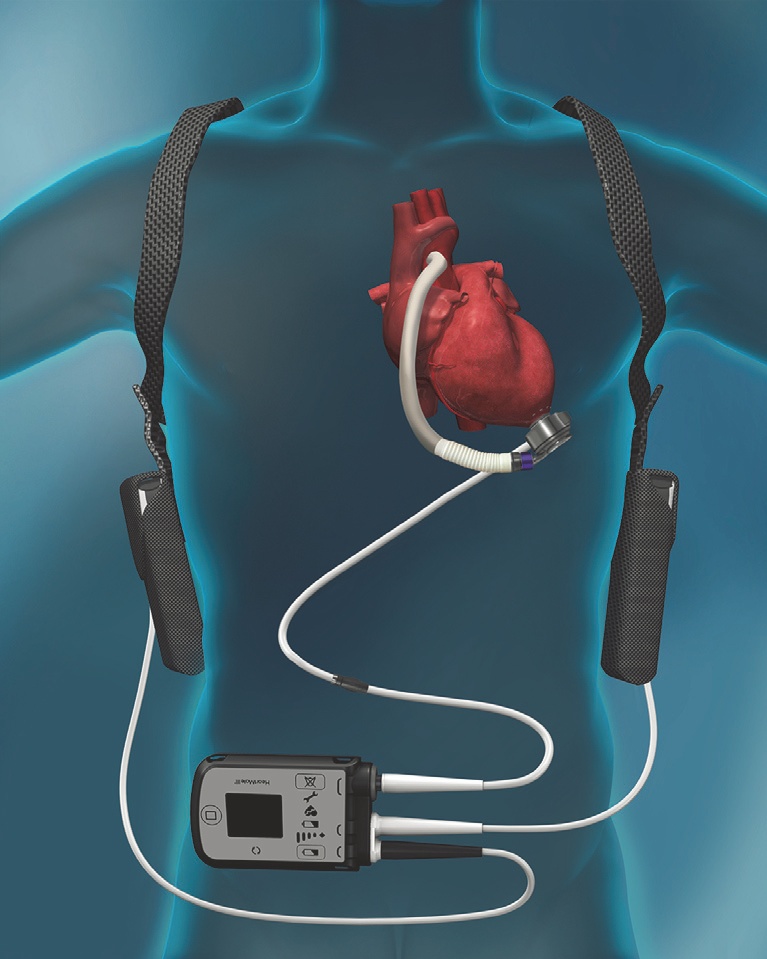
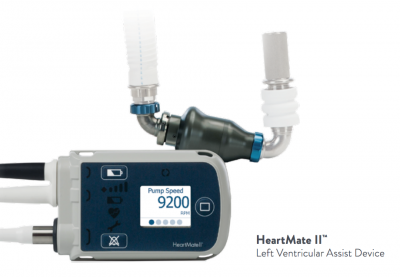
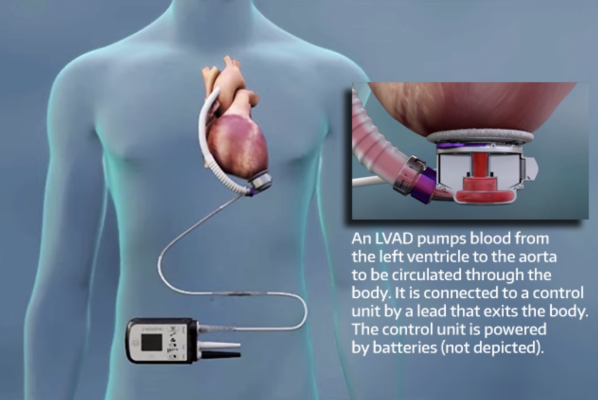
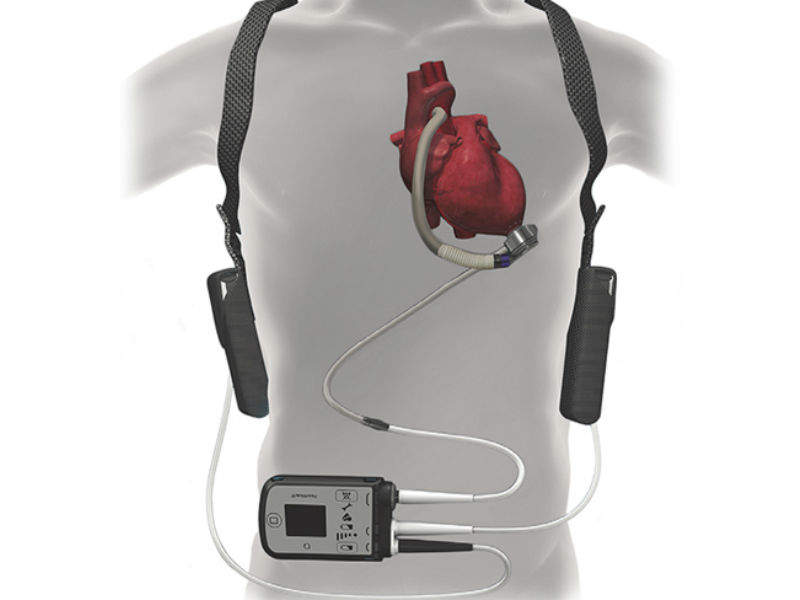
FDA Approves Less Invasive Surgical Approach for Abbott's Heart Pump to Help Patients Avoid Open Heart Surgery - OR Today

New Data Show Abbott's HeartMate 3™ Heart Pump Extends Life Beyond Five Years for Advanced Heart Failure Patients - Aug 29, 2022

Abbott's Heartmate 3™ Heart Pump Extends Life Beyond 5 Years for Advanced Heart Failure Patients – Regenerative Medicine at the McGowan Institute

New Data Show Abbott's HeartMate 3™ Heart Pump Extends Life Beyond Five Years for Advanced Heart Failure Patients - Aug 29, 2022